Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 and GeO
and GeO liquids
liquidsKawaguchi, Munemichi; Uno, Masayoshi*
Journal of Crystal Growth, 585, p.126590_1 - 126590_7, 2022/05
Phase-field mobility,  , and crystal growth rates in crystallization of 11 oxides or mixed oxides in undercooled silicates, SiO
, and crystal growth rates in crystallization of 11 oxides or mixed oxides in undercooled silicates, SiO and GeO
and GeO liquids were calculated with a simple phase-field model (PFM), and material dependence of the
liquids were calculated with a simple phase-field model (PFM), and material dependence of the  was discussed. Ratios between experimental crystal growth rates and the PFM simulation with
was discussed. Ratios between experimental crystal growth rates and the PFM simulation with 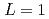 were confirmed to be proportional to a power of
were confirmed to be proportional to a power of 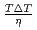 on the solid/liquid interface process during the crystal growth in a log-log plot. We determined that parameters,
on the solid/liquid interface process during the crystal growth in a log-log plot. We determined that parameters, 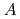 and
and  , of the
, of the 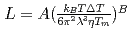 were
were 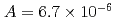 to
to 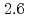 m
m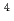 J
J s
s and
and 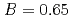 to
to 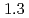 , which were unique for the materials. It was confirmed that our PFM simulation with the determined
, which were unique for the materials. It was confirmed that our PFM simulation with the determined  reproduced quantitively the experimental crystal growth rates. The
reproduced quantitively the experimental crystal growth rates. The 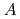 has a proportional relationship with the diffusion coefficient of a cation molar mass average per unit an oxygen molar mass at
has a proportional relationship with the diffusion coefficient of a cation molar mass average per unit an oxygen molar mass at 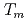 in a log-log graph. The
in a log-log graph. The  depends on the sum of the cation molar mass per the oxygen molar mass,
depends on the sum of the cation molar mass per the oxygen molar mass, 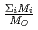 , in a compound. In
, in a compound. In 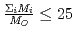 , the
, the  decreases with the cation molar mass increasing. The assumed cause is that the B represents the degree of the temperature dependence of the
decreases with the cation molar mass increasing. The assumed cause is that the B represents the degree of the temperature dependence of the  . Since the cation molar mass is proportional to an inertial resistance of the cation transfer, the
. Since the cation molar mass is proportional to an inertial resistance of the cation transfer, the  decreases with inverse of the cation molar mass. In crystallization of the silicates of heavy cation in
decreases with inverse of the cation molar mass. In crystallization of the silicates of heavy cation in 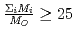 , the
, the  saturates at approximately 0.67, which leads to
saturates at approximately 0.67, which leads to 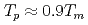 .
.
 O-2SiO
O-2SiO , Na
, Na O-2SiO
O-2SiO and K
and K O-2SiO
O-2SiO
Kawaguchi, Munemichi; Uno, Masayoshi*
Journal of the Ceramic Society of Japan, 128(10), p.832 - 838, 2020/10
Times Cited Count:2 Percentile:16.44(Materials Science, Ceramics)This study developed phase-field method (PFM) technique in oxide melt system by using a new mobility coefficient ( ). The crystal growth rates (
). The crystal growth rates (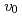 ) obtained by the PFM calculation with the constant
) obtained by the PFM calculation with the constant  were comparable to the thermodynamic driving force in normal growth model. The temperature dependence of the
were comparable to the thermodynamic driving force in normal growth model. The temperature dependence of the  was determined from the experimental crystal growth rates and the
was determined from the experimental crystal growth rates and the 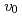 . Using the determined
. Using the determined  , the crystal growth rates (
, the crystal growth rates (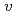 ) in alkali disilicate glasses, Li
) in alkali disilicate glasses, Li O-2SiO
O-2SiO , Na
, Na O-2SiO
O-2SiO and K
and K O-2SiO
O-2SiO were simulated. The temperature dependence of the
were simulated. The temperature dependence of the 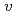 was qualitatively and quantitatively so similar that the PFM calculation results demonstrated the validity of the
was qualitatively and quantitatively so similar that the PFM calculation results demonstrated the validity of the  . Especially, the
. Especially, the 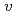 obtained by the PFM calculation appeared the rapid increase just below the thermodynamic melting point (
obtained by the PFM calculation appeared the rapid increase just below the thermodynamic melting point (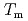 ) and the steep peak at around
) and the steep peak at around 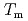 -100 K. Additionally, as the temperature decreased, the
-100 K. Additionally, as the temperature decreased, the 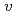 apparently approached zero ms
apparently approached zero ms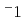 , which is limited by the
, which is limited by the  representing the interface jump process. Furthermore, we implemented the PFM calculation for the variation of the parameter
representing the interface jump process. Furthermore, we implemented the PFM calculation for the variation of the parameter  in the
in the  . As the
. As the  increased from zero to two, the peak of the
increased from zero to two, the peak of the 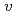 became steeper and the peak temperature of the
became steeper and the peak temperature of the 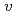 shifted to the high temperature side. The parameters
shifted to the high temperature side. The parameters 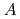 and
and  in the
in the  increased exponentially and decreased linearly as the atomic number of the alkali metal increased due to the ionic potential, respectively. This calculation revealed that the
increased exponentially and decreased linearly as the atomic number of the alkali metal increased due to the ionic potential, respectively. This calculation revealed that the 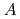 and
and  in the
in the  were close and reasonable for each other.
were close and reasonable for each other.